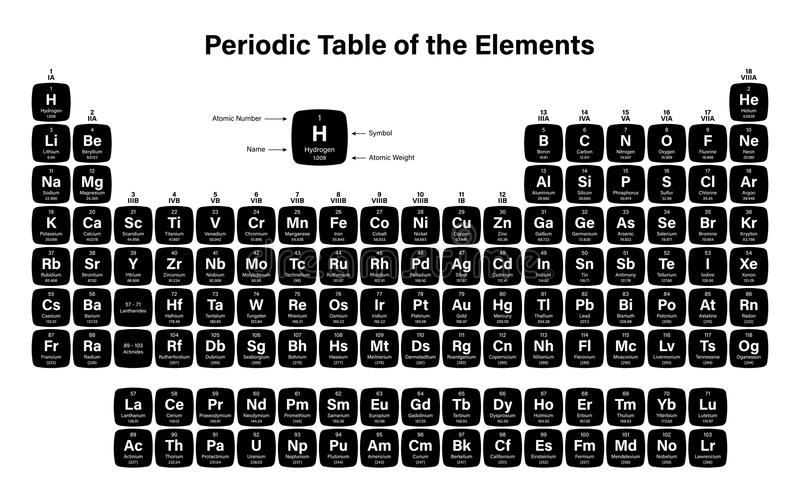
Periodic Table Of Elements With Atomic Mass And Valency And Electronic Configuration, Name Of Elements With Atomic Number Atomic Mass Valency Science Notes Biology Notes Chemistry Notes
Periodic table of elements with atomic mass and valency and electronic configuration Indeed recently has been hunted by consumers around us, perhaps one of you personally. People now are accustomed to using the net in gadgets to see image and video information for inspiration, and according to the title of the article I will talk about about Periodic Table Of Elements With Atomic Mass And Valency And Electronic Configuration.
- Structure Of The Atom Chapter Notes Dronstudy Com
- Ch150 Chapter 2 Atoms And Periodic Table Chemistry
- Periodic Table Groups Periods Trends Patterns Comparison Properties Of Metals Non Metals Allotropes Summary Overview Gcse Chemistry Revison Notes Igcse O Level Ks4 Science
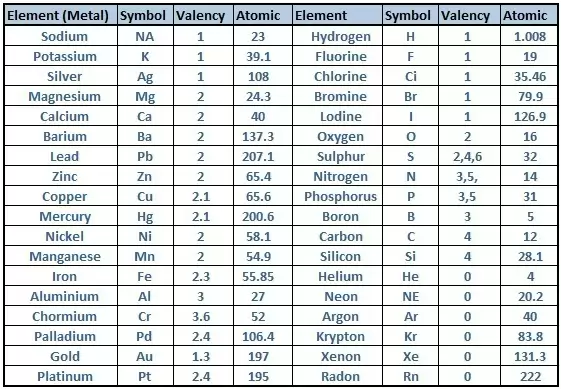
- Periodic Table Of Elements With Atomic Mass And Valency Pdf Download
- Periodic Classification Of Elements Class 10 Notes Science Chapter 5 Msr Blog
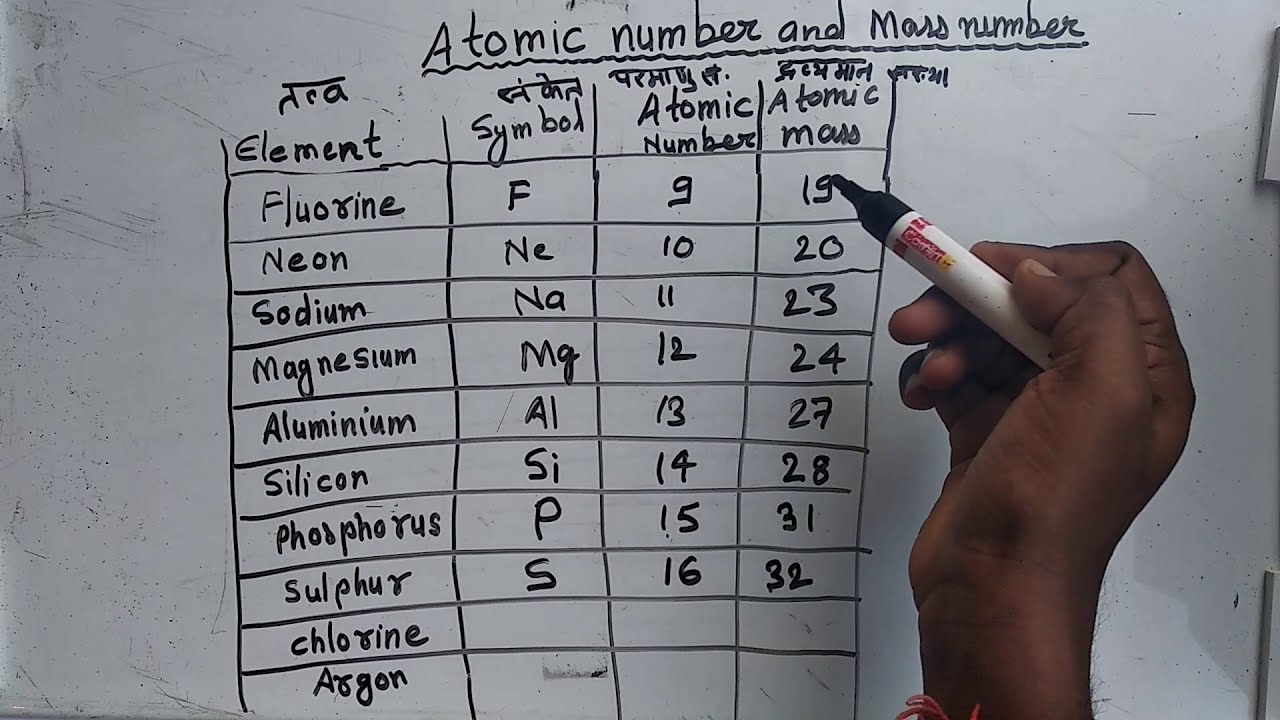
- What Are The First 20 Elements Names And Symbols
Find, Read, And Discover Periodic Table Of Elements With Atomic Mass And Valency And Electronic Configuration, Such Us:
- Color Periodic Table Of The Elements Valence Charge
- Exam Point Of View Questions 2016
- 9 7 Electron Configurations And The Periodic Table Chemistry Libretexts
- Course S1 Chemistry Topic Unit 5 Atoms Elements And Compounds
- Https Encrypted Tbn0 Gstatic Com Images Q Tbn 3aand9gcrlck V9ct Rmf8qcsxirfrpd67xbspwtwfzkkh Dj4sd88 Yf Usqp Cau
If you are searching for Autumn Wreath Craft you've come to the perfect place. We ve got 104 images about autumn wreath craft adding pictures, photos, pictures, backgrounds, and more. In such page, we also have number of images out there. Such as png, jpg, animated gifs, pic art, logo, black and white, transparent, etc.
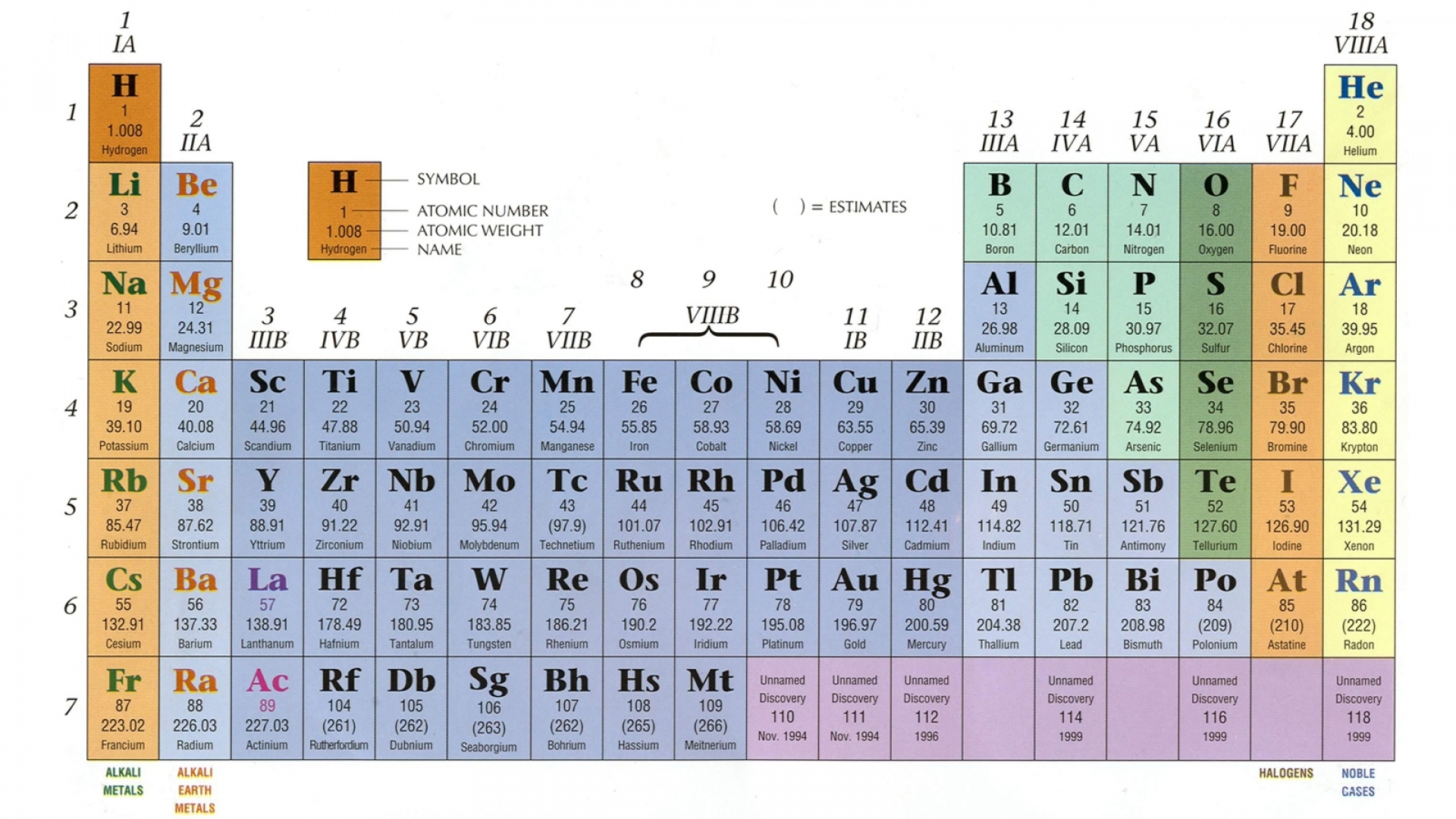
This information is available on a color periodic table of the elements or a black and white version.
Autumn wreath craft. Thus in the building up process for the lanthanoids. Electronic configuration of first 30 elements with atomic numbers. Knowledge of the electron configuration of different atoms is useful in understanding the structure of the periodic table of elements.
There are 118 elements in the periodic table. Hydrogen is a chemical element with atomic number 1 which means there are 1 protons and 1 electrons in the atomic structurethe chemical symbol for hydrogen is h. The modern periodic table is based on the modern periodic law put forward by the english physicist henry moseley which states that the properties of.
You may assume that the valences of the elementsthe number of electrons with which an atom will bond or formare those that can be derived by looking at the groups columns of the periodic table. The electron configuration is the distribution of electrons of an atom or molecule or other physical structure in atomic or molecular orbitals. Variation of oxidation state along a period.
Its monatomic form h is the most abundant chemical substance in the universe constituting roughly 75 of all baryonic mass. The periodic table is a tabular display of the chemical elements organized on the basis of their atomic numbers electron configurations and chemical properties. Each element has a unique atomic structure that is influenced by its electronic configuration which is the distribution of electrons across different orbitals of an atom.
The first example occurs in the case of the lanthanoids elements having atomic numbers between 57 and 71the lanthanoids have the general electron configuration kr4d 10 4f i 5s 2 5p 6 5d 0 or 1 6s 2. But the valency of elements when combined with h or o first increases from 1 to 4 and then it reduces to zero. Periodic table 1864 john newlands law of octaves every 8th element had similar properties when arranged by atomic masses not true past ca 1869 dmitri mendeleev lothar meyer independently proposed idea of periodicity recurrence of properties.
For example the electronic configuration of sulfur can be written as ne 3s 2 3p 4 since neon has an electronic configuration of 1s 2 2s 2 2p 6. In the periodic table the vertical columns are called groups and the horizontal rows are called periods. While moving left to right across a period the number of valence electrons of elements increases and varies between 1 to 8.
The bond valence model 2nd ed. The periodic table is the tabular arrangement of all the chemical elements on the basis of their respective atomic numbers. Given below is a table describing the electronic configuration of first 30 elements with atomic numbers.
A simpler version listing only the most common valence charges is also available. With a standard atomic weight of circa 1008 hydrogen is the lightest element on the periodic table. Periodic table with element valences.
The third major category of elements arises when the distinguishing electron occupies an f subshell. Periodic trends in the oxidation states of elements 1. Where i is a number between 0 and 14.
More From Autumn Wreath Craft
- Unique Aesthetic Baddie Wallpapers Orange
- Differenza Tra Pistola A Tamburo E Automatica
- Pink Aesthetic Pictures Baddie Quotes
- Womens Autumn Winter Outfits
- Ps5 Price Reveal Date
Incoming Search Terms:
- The History Of The Periodic Table Boundless Chemistry Ps5 Price Reveal Date,
- Color Periodic Table Of The Elements Valence Charge Ps5 Price Reveal Date,
- Free Printable Periodic Tables Pdf Ps5 Price Reveal Date,
- Periodic Table Of Elements With Atomic Mass Ps5 Price Reveal Date,
- Discovering Chemistry 3 2 Valency The Combining Power Of An Atom Openlearn Open University Dc 1 Ps5 Price Reveal Date,
- 118 Elements And Their Symbols Atomic Mass Or Valency Ps5 Price Reveal Date,
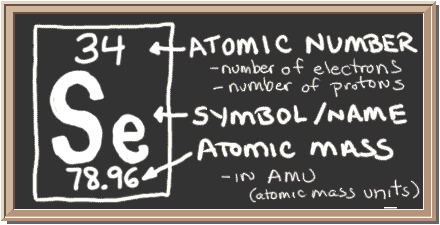
/what-are-the-first-20-elements-608820-FINAL-5b758ab446e0fb002c67279a.png)
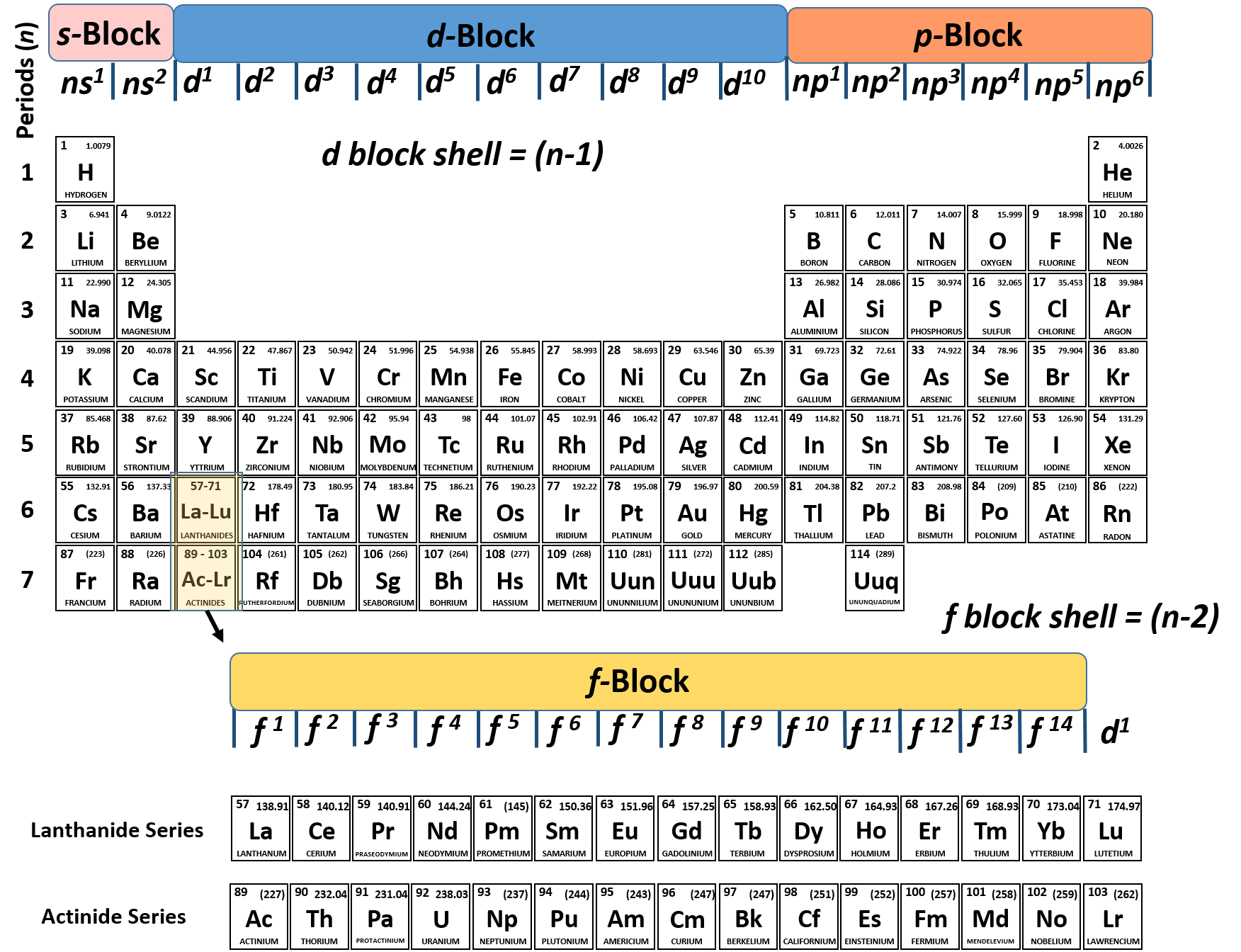

/186810031-56a130cd5f9b58b7d0bce8ee.jpg)